Contract Manufacturing
Complete solutions at any stage of the development and manufacturing process / Complete solutions for manufacturers including design, production, assembly, and distribution.
Capabilities
Benefit from industry-leading expertise in precision dispensing for human and veterinary diagnostics, food and environmental monitoring, life science institutions, and Pharma and MedTech companies. Our production batches range from as little as xyz to as many as 1xyz per day. Drop-on-demand dispensing of
10 pL to 100 μL can be covered by a single multiplex dispensing platform.
This range can be extended based on customer requirements.

Bridge the gap from research to commercialization.
We’ll help you accelerate product development and transform your ideas into products.
Complete solutions for your workflow
Reduce your spend on costly investments to bring your product to market. We provide all requirements for your workflow, including equipment, resources, facilities, and ISO certification.

Go from proof-of-concept to high-throughput production with a single partner.
SCIENION’s experienced team of assay developments experts will translate research-scale innovations into certified manufacturing systems.
Adaptable and flexible to your volume needs
We understand that things change. We can be flexible and adapt to your volume needs to reduce your risk. (needs edit)
Add background: cellenCHIP image
Decide later between contract manufacturing and in-house production
Start production immediately with SCIENION with the option for tech transfer later if required. We’ll support you through every step, at our facilities or yours.
ISO Certified
SCIENION AG and SCIENION (UK) Ltd are both certified according to DIN EN ISO 9001:2015 regarding development, manufacture and sales of dispensing systems, diagnostic manufacturing devices and microarrays.

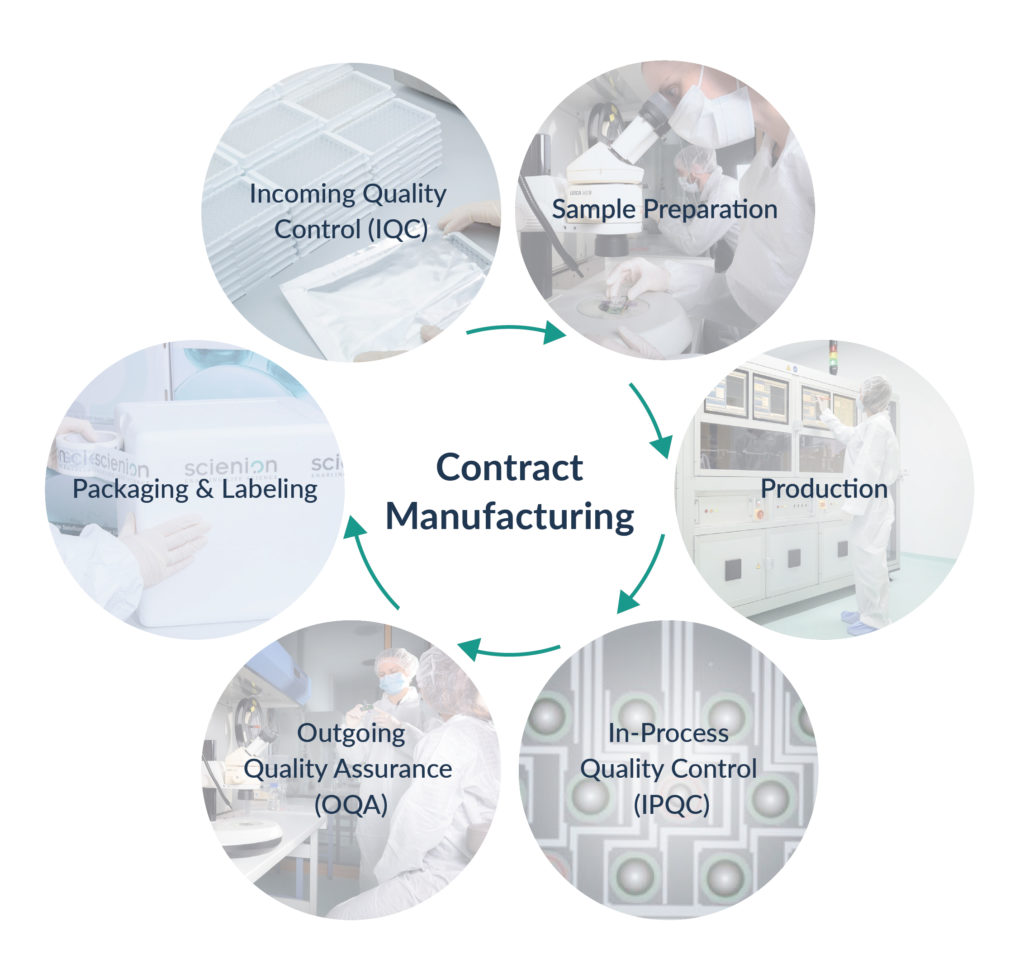
Quality Control & Assurance
- DIN EN ISO 9001:2015 Quality management system
- CE marked instrumentation for high throughput production
- Dust minimized and controlled production environment (HEPA filter)
- Advanced in-process QC, 100% automated array control
- Functional QC: Application tests of randomly selected arrays
- QC certificate provided alongside with each batch of arrays
Probes
Supports


Diagnostic Test Features
Full Service Range
- Supply of materials and preparation of source plates
- Microarray printing and processing (immobilization, blocking, washing)
- Quality control of supports, array parameters and array functionality
- Electronic documentation of the production process
- Packaging according to customers requirements
- OEM microarray production